Drug Repurposing Approaches Reveal HIV Drug Holds Promise Against a Rare, Disabling Muscle Disorder
November 30, 2021
Dystonia — a group of rare, potentially disabling muscle disorders characterized by abnormal, involuntary movements — has few treatments and no cure. Drugs and other therapies, such as deep brain stimulation, may help, but they treat only the symptoms.
A collaborative research team from Duke University, the University of Florida and NCATS has shown that an existing antiviral drug used against HIV infection may prove helpful in treating DYT1, the most common inherited form of dystonia. Scientists discovered that the drug ritonavir could correct the results of a genetic mistake that causes DYT1. The promising results, seen both in cell culture and in an animal model of the disease, suggest the drug or similar versions could be further developed as potential therapies for dystonia. The researchers reported their findings in Science Translational Medicine.
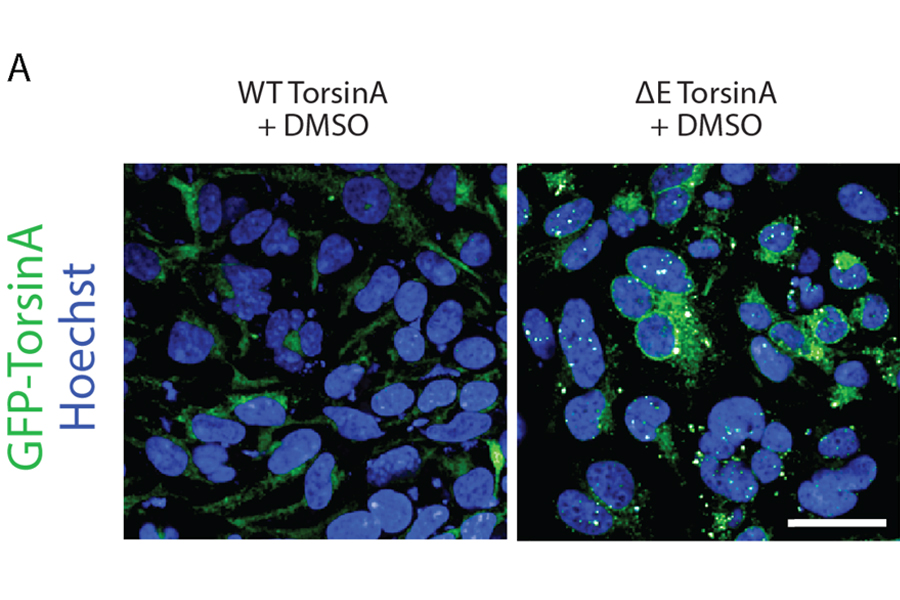
DYT1 is caused by a genetic mutation that produces an abnormal protein. The defective protein disrupts communication in nerve cells that control muscle movement. Several years ago, Duke neurologist and co-author Nicole Calakos, M.D., Ph.D., and her team developed an assay, or test, to gauge the ability of drugs to correct the protein defect. But they could not find a compound or drug that would work well enough. They reached out to NCATS scientists to collaborate and take advantage of the Center’s drug screening and repurposing expertise, along with its vast drug and compound collections. Drug repurposing, which entails using existing drugs in a new, unintended way, can be a helpful strategy to treat diseases.
To find a drug that could be used to treat the disease, NCATS translational scientists, led by Min Shen, Ph.D., and Matt Hall, Ph.D., assessed the effectiveness of the nearly 3,000 U.S. Food and Drug Administration–approved drugs in the NCATS Pharmaceutical Collection. They adapted the assay to be able to use high-throughput screening to test the collection rapidly and found 18 promising drugs, including several antiviral drugs known as HIV protease inhibitors. Ritonavir appeared the most effective in correcting the protein abnormality.
“Adapting the assay for screening NCATS’ drug library was a key to identifying drug repurposing opportunities,” said Shen. “Once we identified the ritonavir as a potentially disease-modifying therapeutic opportunity for dystonia, Duke scientists demonstrated that the drug was effective.”
Using the DYT1 mouse models they developed, Calakos and her co-workers showed that the drug could correct the protein defect and related abnormalities in adult mouse brain tissue from mice with the DYT1 gene mutation.
In a second experiment, the researchers compared young DYT1 mice that received the drug daily for two weeks to DYT1 mice that did not receive the drug. After imaging the mouse brains in adulthood, the researchers found that brains of mice with the disorder that received ritonavir looked the same as healthy control mice.
“The deep expertise within the drug screening and development teams at NCATS was indispensable to our work,” said Calakos. “We now have the first suggestion from animal models that disease-modifying treatments might be possible. Our top priority looking ahead is to find ways to get this drug or related compounds into the human brain at the levels needed so that we can test its safety.”
The researchers believe the antiviral drug, and possibly other similar drugs, work through an unintended, “off-target” effect. They think the drug affects how cells respond to stress in DYT1. This stress response could play a role in multiple diseases, Shen noted. For now, Duke and NCATS researchers will work to develop more effective, alternate forms of ritonavir, with a goal of eventually finding new therapies for dystonia in people.
“We will continue to explore ritonavir as a treatment possibility,” Shen said, “and also study how it works to drive the development of new treatments for this rare disease.”