NIH Awards $89 Million for Additional Projects to Advance Genome Editing
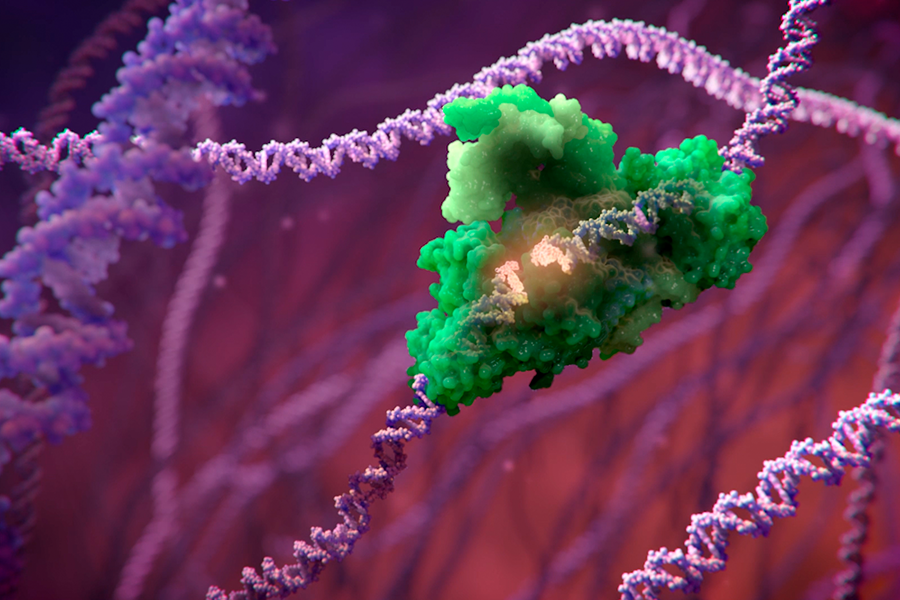
An illustration depicting an editing tool repairing a DNA strand. Some gene-editing tools use enzymes to cut the disease-causing DNA and bind the unchanged sequences back together. (XVIVO)
September 30, 2019
Funding aimed at developing new technologies, novel research approaches in the Somatic Cell Genome Editing Program
Genome editing is a promising technology that could offer new treatments or cures for diseases, but challenges remain. To help address them, the National Institutes of Health (NIH) has awarded 24 additional grants to researchers across the United States and Canada through the Somatic Cell Genome Editing (SCGE) Program. This group of awards from the SCGE Program totals approximately $89 million over four years. This brings the total number of projects supported to 45 and total funding to approximately $190 million over six years, pending available funds.
The human genome contains thousands of genes, each of which contains the information that cells use to make proteins. In genetic diseases, there is a problem with a gene such that a cell may not make a protein, or it may make a protein incorrectly. Genome editing aims to change the DNA sequence so that cells can make the right proteins again. NIH created the SCGE Program in January 2018 to improve genome-editing technologies and make genome-editing therapies more widely available. All the projects focus on somatic (nonreproductive) cells in the body, which do not pass on DNA changes to future generations. As a result, the changes made through somatic cell genome editing — which can carry both risks and benefits — cannot be inherited. The program is supported by the NIH Common Fund and led by NCATS. NCATS announced the first group of SCGE grants last October.
“Genome editing has extraordinary potential to alter the treatment landscape for common and rare diseases,” said Christopher P. Austin, M.D., NCATS director and SCGE Program Working Group chair. “The field is still in its infancy, and these newly funded projects promise to improve strategies to address a number of challenges, such as how best to deliver the right genes to the correct places in the genome efficiently and effectively. Together, the projects will help advance the translation of genome-editing technologies into patient care.”
The initial group of SCGE grantees is beginning to show research results. For instance, University of Wisconsin scientists recently reported the development of a tiny “nanocapsule” to deliver gene-editing tools to targeted cells more accurately and effectively than before. Information about SCGE approaches, technologies and tools will be disseminated in a toolkit through the SCGE dissemination and coordinating center.
“NIH Common Fund programs are meant to have exceptional impact across a broad area of science,” said Elizabeth Wilder, Ph.D., director of NIH’s Office of Strategic Coordination, which oversees the Common Fund. “As a Common Fund program, SCGE aims to transform the field of genome editing by developing new tools and moving them toward clinical use.”
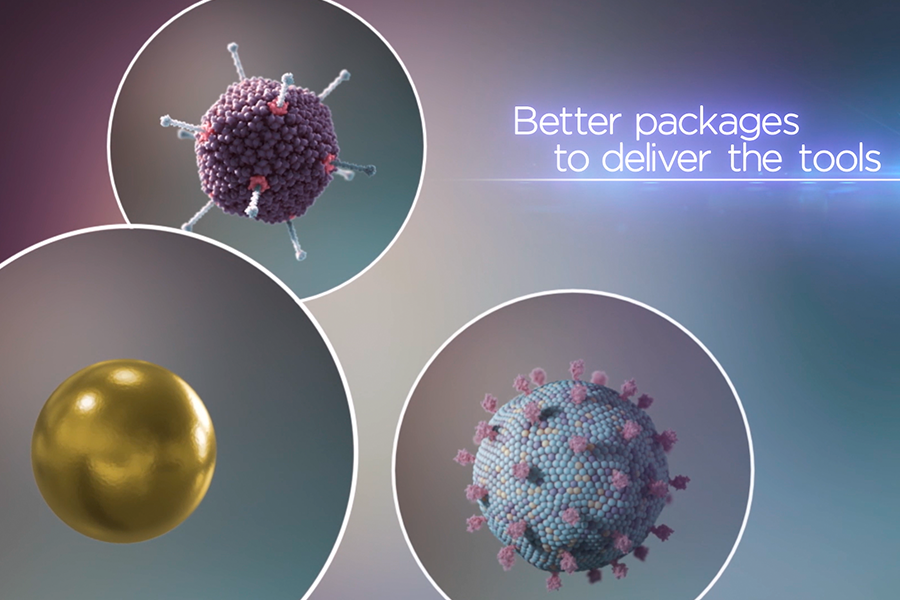
An illustration depicting tailored delivery vehicles that are packaged with gene-editing tools. These delivery techniques must be specific to enter only the cells with the disease-causing DNA and avoid the unaffected ones. (XVIVO)
New Awards
The newly funded projects expand the number of target cells and tissues, as well as the types of genome-editing technologies under investigation. One project, for example, will assess the ability of nanoparticles to deliver editing tools to up to 30 different types of target tissues. This will be the first SCGE project to use zinc finger nuclease technology as an editing tool. This technology, which differs from the better known CRISPR-Cas9 technique, already has been used in patients in a clinical trial to treat a rare metabolic disease.
A different initiative will develop tools and technologies that will enable monitoring and tracking of genome-edited cells in humans to better assess the safety and efficacy of genome-editing therapies.
Another new award supports the development of an epigenome editor, which can regulate how genes are expressed in target cells. And whereas most genome-editing tools use enzymes as molecular scissors to make specific cuts in DNA, two projects, including one that is newly funded, use small pieces of modified DNA to trigger the cell’s natural abilities to repair damaged DNA and edit the genome without cutting the DNA.
“The DNA repair approach is very different from better-known genome-editing methods, such as CRISPR-Cas9,” said P. J. Brooks, Ph.D., program director for the NCATS Office of Rare Diseases Research and SCGE program coordinator. “When we developed the program, we felt that it was important to include a diversity of technologies and approaches.”
Brooks also emphasized the inclusion of independent testing centers within the SCGE to test and validate the new delivery technologies developed in the program. He noted that such independent validation will accelerate translation of these technologies into clinical use.
In addition to NCATS, other NIH Institutes and Centers managing these awards include the National Heart, Lung, and Blood Institute; National Institute of Allergy and Infectious Diseases; National Institute of Neurological Disorders and Stroke; National Institute of Arthritis and Musculoskeletal and Skin Diseases; National Institute of Biomedical Imaging and Bioengineering; National Human Genome Research Institute; and the Office of Research Infrastructure Programs.
For more information about somatic cell genome editing, visit ncats.nih.gov/research/research-activities/scge. For a list of program grantees and information about their awards, see commonfund.nih.gov/editing/fundedresearch.


