NCATS Pharmaceutical Collection
The NCATS Pharmaceutical Collection is a comprehensive, publicly accessible collection of approved molecular entities for high-throughput screening that provides a valuable resource for both validating new models of disease and better understanding the molecular basis of diseases and interventions.
About the NCATS Pharmaceutical Collection
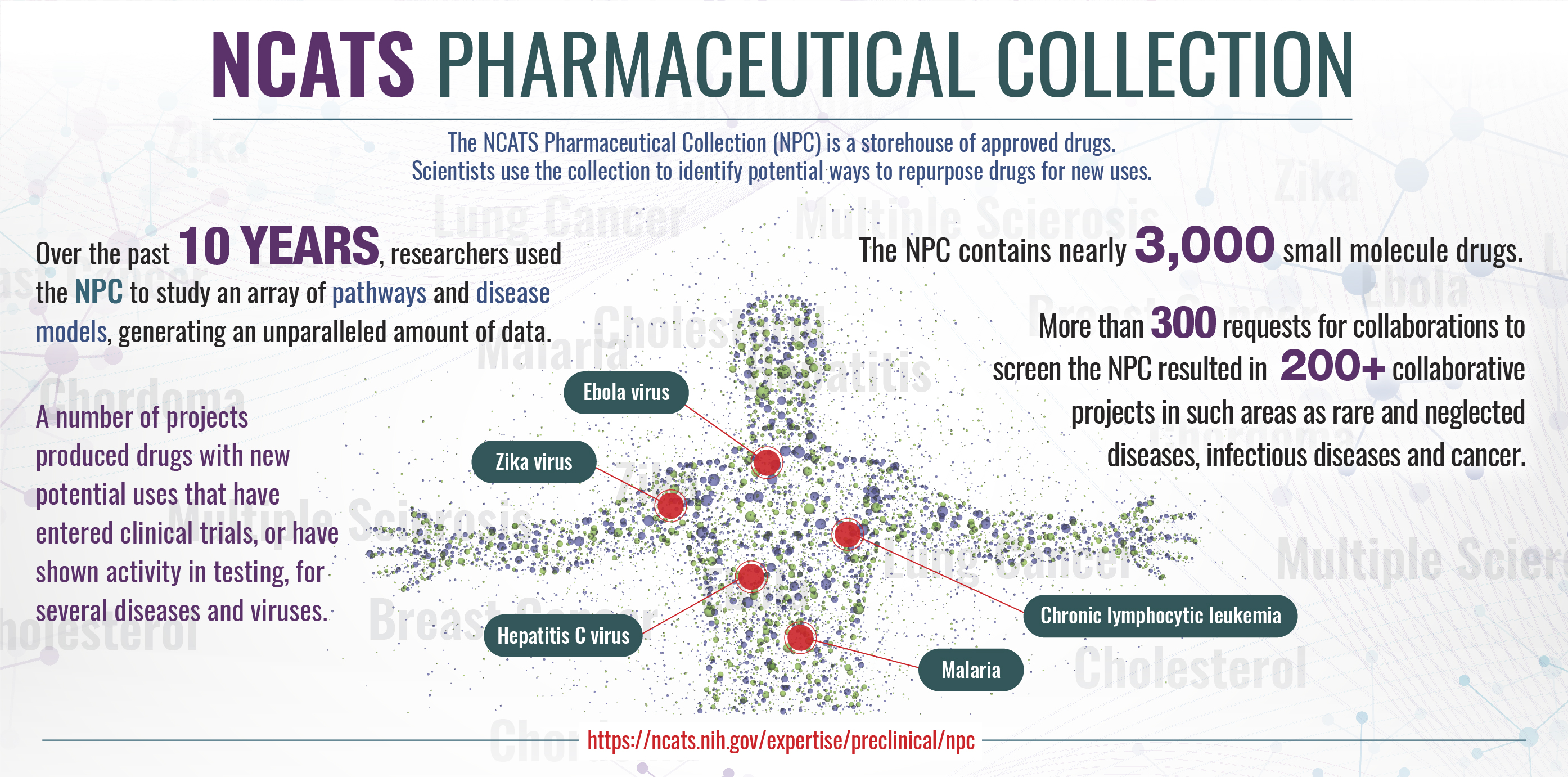
Nearly 3,000 small molecular entities have been approved for clinical use by U.S., European Union, Japanese, Australian and Canadian authorities and also are suitable for high-throughput screening. Of these, we currently have 2,900 as part of our screening collection.
The collection already has generated several useful probes for studying a diverse cross section of biology, including novel targets and pathways. We provide access to the collection’s content through the Therapeutics for Rare and Neglected Diseases program and the Toxicology in the 21st Century initiative. A dedicated online browser enables searching and exporting. We make regular updates to the browser to improve features and content. Download the latest version.
Sources for the current collection include traditional chemical suppliers, specialty collections, pharmacies and custom synthesis. All data generated through this effort are deposited in PubChem, including the full concentration-response profile of each compound in each assay. Detailed information on the sourcing of the collection, including regulatory status, supplier information, compound structures, target information and indication, are made available as time permits.
We developed a new OpenData Portal to share COVID-19-related drug repurposing data and experiments for all approved drugs. To develop the portal, researchers used SARS-CoV-2-related assays to screen over 10,000 compounds, including the compounds from the NCATS Pharmaceutical Collection, for their activity against the virus.
View the infographic below to learn more about the NCATS Pharmaceutical Collection. The NCATS Pharmaceutical Collection is described in more detail in a recent publication and an NCATS Director’s Message.
(NCATS)