NIH expands clinical trials to test convalescent plasma against COVID-19
UPDATE:
- Results from the CONTAIN COVID-19 trial showed that for people hospitalized with COVID-19, convalescent plasma taken from those who had recovered was not more effective than placebo in delivering clinical improvement. Read a statement about the results published in January 2022.
- Results from the Passive Immunity Trial for our Nation (PassITON) showed that treatment with convalescent plasma did not improve clinical outcomes in adults hospitalized with COVID-19. Read the journal article published in June 2022.
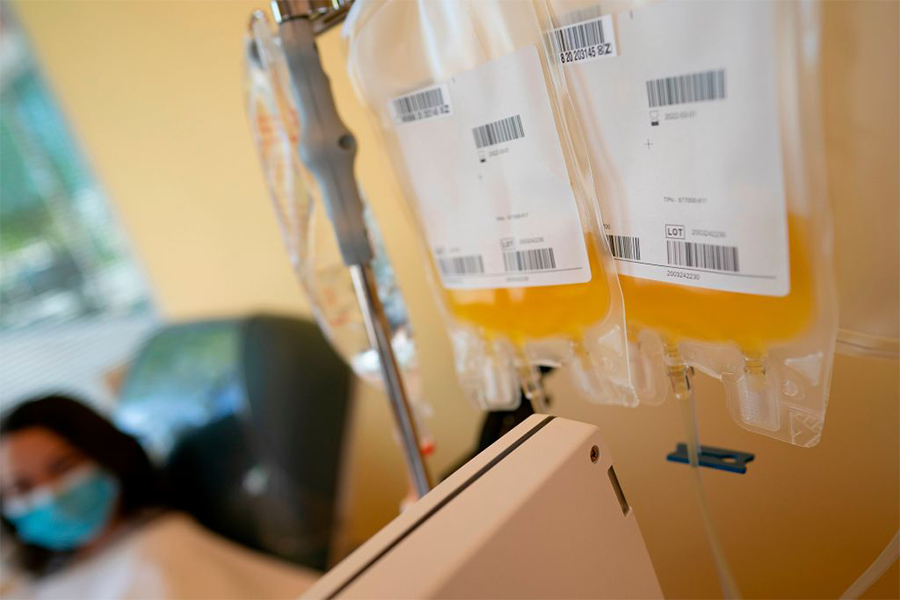
Two randomized, placebo-controlled clinical trials are evaluating convalescent plasma from people who have recovered from COVID-19 as a treatment for those hospitalized with COVID-19. (Alex Edelman/AFP via Getty Images)
September 22, 2020
Rigorous studies to build on earlier efforts to test the experimental treatment
Two randomized, placebo-controlled clinical trials funded by the National Institutes of Health (NIH) are expanding enrollment to further evaluate convalescent plasma as a treatment for patients hospitalized with COVID-19. Preliminary observational studies indicate that convalescent plasma may improve outcomes among severely ill and hospitalized patients with COVID-19. Prospective, well-controlled randomized trials are needed to generate sufficient data on whether convalescent plasma is effective and safe for the treatment of COVID-19.
Convalescent plasma is blood plasma taken from people who have recovered from COVID-19. It contains antibodies that can recognize and neutralize SARS-CoV-2, the virus that causes COVID-19, as well as other components that may contribute to an immune response.
“The evidence on convalescent plasma as a treatment for severe cases of COVID-19 is promising but incomplete. We need to carry out rigorous randomized control clinical trials to determine how this therapy can improve outcomes,” said NIH Director Francis S. Collins, M.D., Ph.D. “While the world waits for an effective vaccine, it is vital that we simultaneously expand the options for available treatments for those currently suffering from the worst effects of this disease.”
The trials expect to enroll hospitalized patients across the country at academic and community-based hospitals. Participants will be randomly assigned to receive the treatment or a placebo. Outcomes will be compared with respect to clinical improvement measures and resource needs, such as ventilators. Both trials currently are enrolling participants and anticipate results as early as this fall.
The trials are receiving $48 million in support through Operation Warp Speed (OWS), a collaborative initiative across federal agencies to advance the development, manufacturing and distribution of COVID‑19 vaccines, therapeutics and diagnostics.
The National Center for Advancing Translational Sciences (NCATS), part of NIH, will oversee the grant awards through its Clinical and Translational Science Awards (CTSA) Program research network. The CTSA’s Trial Innovation Network (TIN) will play a key role in working to add study sites and enroll patients, including those from communities disproportionately affected by COVID-19.
“The rapid expansion of these vital randomized, controlled convalescent plasma clinical trials demonstrates how nimbly the network of CTSA Program hubs and the TIN can respond to the nation’s research needs and shorten the path from discovery to treatment,” said NCATS Director Christopher P. Austin, M.D.
One trial, called Convalescent Plasma to Limit COVID-19 Complications in Hospitalized Patients, was launched in April by NYU Langone Health in New York, with collaboration from the Albert Einstein College of Medicine and Yale University, New Haven, Connecticut, and with funding from NCATS. To increase enrollment in the trial, NYU is partnering with The University of Texas Health Science Center at Houston and the University of Miami in Florida to enroll participants at sites in these states.
With these additional sites, this trial expects to enroll approximately 1,000 hospitalized patients 18 years or older with respiratory symptoms of COVID-19. The trial is primarily assessing clinical improvement at 14 and 28 days and also will be evaluating outcomes based on mortality, intensive care unit admission and patient antibody concentrations. Additional information about this study and participation is available at ClinicalTrials.gov under study identifier NCT04364737.
The trial called Passive Immunity Trial of Our Nation for COVID-19 also is expanding to enroll about 1,000 participants. Vanderbilt University Medical Center in Nashville, Tennessee, which launched the trial in April, will have access to about 50 additional clinical trial sites across the CTSA Program. Participants are 18 years or older with acute respiratory infection symptoms and laboratory-confirmed SARS-CoV-2 infection; they may be hospitalized or in an emergency department and likely to be admitted. The trial primarily will assess clinical improvement at 15 days and also will evaluate ventilation use, supplemental oxygen use, acute kidney injury and cardiovascular events. Additional information about this study and participation is available at ClinicalTrials.gov under study identifier NCT04362176.
Media Contact: info@ncats.nih.gov
About the National Center for Advancing Translational Sciences (NCATS): NCATS conducts and supports research on the science and operation of translation — the process by which interventions to improve health are developed and implemented — to allow more treatments to get to more patients more quickly. For more information about how NCATS helps shorten the journey from scientific observation to clinical intervention, visit https://ncats.nih.gov.
About the National Institutes of Health (NIH): NIH, the nation’s medical research agency, includes 27 Institutes and Centers and is a component of the U.S. Department of Health and Human Services. NIH is the primary federal agency conducting and supporting basic, clinical, and translational medical research, and is investigating the causes, treatments, and cures for both common and rare diseases. For more information about NIH and its programs, visit https://www.nih.gov.
NIH…Turning Discovery Into Health®


