MMA-101
The MMA-101 gene therapy project will offer new hope for people living with rare diseases.
About MMA-101 Gene Therapy Trial
NCATS and NIH’s National Human Genome Research Institute (NHGRI) are collaborating to test a gene therapy as a potential treatment for a rare genetic disorder of the liver called methylmalonic acidemia (MMA). The gene therapy treatment will deliver a working copy of a faulty gene that is needed to break down fats and proteins.
MMA prevents the body from breaking down fats and proteins properly. This causes a buildup of acids and other harmful substances in the blood, urine and cells, including brain cells. Long-term complications of MMA include developmental delays, intellectual disability and chronic kidney disease.
This trial will use a common gene therapy vector, adeno-associated virus 8 (AAV8), to deliver a healthy copy of the methylmalonyl-CoA mutase (MMUT) gene to a small number of children aged 3-18 with the disease. The study is designed as a Phase 1/2 trial and will allow researchers to see if the therapy is safe and shows some signs of treating the disease. The gene therapy treatment may slow the progression of the disease and provide an alternative treatment option for the liver and kidney transplants that people with recurrent and severe MMA receive.
MMA-101 could inform the Platform Vector Gene Therapy (PaVe-GT) pilot and the Bespoke Gene Therapy Consortium (BGTC), both of which have gene therapy clinical trial projects in development. It also could help further establish a scientific and operational framework for gene therapy trials for rare diseases at the NIH Clinical Center.
NCATS is committing about $2.2 million over five years to the project through its Cures Acceleration Network. Other NIH institutes and centers, such as the National Institute of Neurological Disorders and Stroke (NINDS) and the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), and the NIH Clinical Center also will provide infrastructure and staffing to support the trial.
MMA-101 News
New Path for a Gene Therapy Trial at NIH for a Rare Metabolic Disease
November 19, 2024 - NCATS News
- Bespoke Gene Therapy Consortium (BGTC)
- Our Impact on Clinical Trials
- Our Impact on Drug Discovery and Development
- Our Impact on Rare Diseases
- Platform Vector Gene Therapy (PaVe-GT)
Scientists at NCATS and other NIH institutes and centers are teaming up to advance a gene therapy treatment for MMA, a rare and potentially life-threatening genetic disorder.
Related Research

Bespoke Gene Therapy Consortium
We help lead this public-private partnership program that focuses on developing platforms and standards to speed the development and delivery of gene therapies for rare diseases.
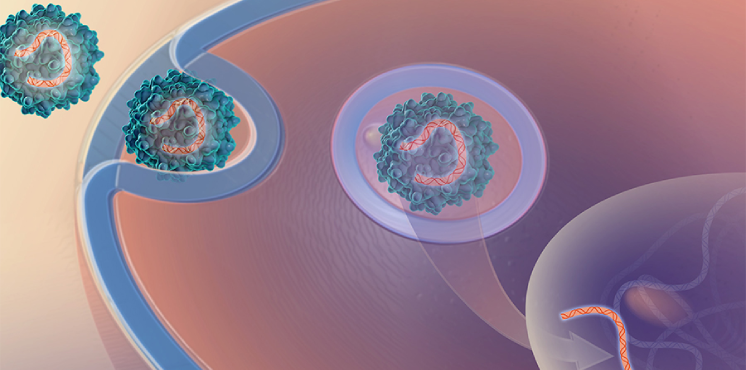
Platform Vector Gene Therapy
Our pilot project is testing the feasibility of increasing the efficiency of gene therapy clinical trial startups by using the same gene delivery system and manufacturing methods for multiple gene therapies.


