Our Impact on Drug Discovery and Development
Our research teams develop and apply cutting-edge approaches that speed therapeutic solutions for unmet health needs.
Moving More Treatments from the Lab to the Clinic
A new drug’s journey from the lab to the medicine cabinet can take up to 15 years. On the way, it passes through dozens of steps — and potential failure points. As a result, many never get approved for human use.
For more than 10 years, we’ve been making the drug discovery and development steps faster and more predictable. We built a pharmaceutical collection of nearly every drug approved for humans. We also built automated solutions to swiftly test drugs or combinations of them for new uses, including emerging infectious diseases. Through our partnerships with academia, industry and patient advocacy groups, we have enabled more than 45 promising new drugs to move into clinical trials.
Our newer drug discovery and development efforts focus on preparing for future pandemics and building a one-stop shop for finding, designing and testing new molecules with therapeutic potential.
Impact Stories
New Screening Tool Could Rapidly Reveal Better Cancer-Drug Combinations
Scientists at NCATS and NCI designed a screening method that could help predict in the lab how combos of three or more cancer drugs will work in people.
NCATS Enables IND Clearances and Drug Approvals

Our labs play a critical, hands-on role in getting new therapies to patients by partnering to discover and develop drugs for rare diseases and other unmet medical needs.
Drug Discovery and Development Research Activities
We speed preclinical research on promising new medicines through a variety of research activities.

Preclinical Chemical Biology Laboratory
Our scientists and collaborators bridge chemistry and the biology underlying disease to develop new ways to discover and create chemical probes and potential drug candidates.
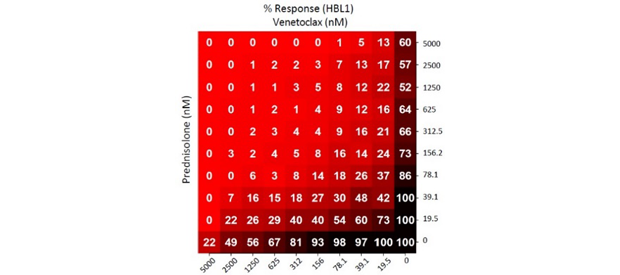
Matrix Combination Screening
Our experts use matrix combination screening technology to quickly identify promising drug combinations with the most potential to help patients.

Compound Management
Our compound management team uses sophisticated and automated techniques to supply chemicals for NCATS screening experiments to uncover new treatments for diseases.
Drug Discovery and Development Research News

FDA Clears IND for Gene Therapy Candidate to Treat Rare Metabolic Disorder
May 21, 2026 - NCATS News
- Our Impact on Clinical Trials
- Our Impact on Drug Discovery and Development
- Our Impact on Rare Diseases
MMA-101, an NIH-supported AAV gene therapy for a rare metabolic disorder, has received FDA clearance to begin clinical testing.
Read ArticleBrigatinib Now in the NCCN Guidelines for NF2-SWN
April 28, 2026 - Grantee/Partner News
- Our Impact on Clinical Trials
- Our Impact on Drug Discovery and Development
- Our Impact on Rare Diseases
How NCATS Is Accelerating Drug Discovery: A Behind-the-Scenes Look With NIH Leadership
April 16, 2026 - NCATS News
- Our Impact on Drug Discovery and Development
NIH senior leadership toured NCATS’ laboratories to learn how the center supports innovative technology to model human disease and explores drug repurposing.
Lupin and TB Alliance Join Forces to Advance Telacebec for the Treatment of Tuberculosis and Other Mycobacterial Diseases
February 2, 2026 - Grantee/Partner News
- Our Impact on Drug Discovery and Development



