Prior Approval Requests for Human Subjects Research
NCATS Process for CTSA Program Prior Approval Requests for Human Subjects Research
Consistent with NOT-OD-15-129, the NCATS process for Clinical and Translational Science Awards (CTSA) Program human subject research (HSR) applies to:
- New UL1/UM1 Projects that involve human subjects research and are supported with direct CTSA grant funding and/or voluntary committed cost share; and/or
- New KL2/K12 Projects that involve human subjects research and are supported with direct CTSA grant funding; and/or
- New UM1 Pilot Projects that involve human subjects research and are supported with direct CTSA grant funding (Reminder: voluntary cost share is not accepted in the CTSA Pilot Module of the UM1 NoFos therefore these pilots must not be submitted for prior approval); and/or
- Delayed onset UM1 Element E Research projects; and/or
- New UM1 Element E Research Projects that were not submitted with the original application
Relevant Notices:
- NOT-TR-21-026: Notice of Information: Change in NCATS Prior Approval Process for New CTSA Program Pilot Projects and KL2 Projects that Involve Human Subjects Research
- NOT-TR-23-007: Notice of Information: NCATS Prior Approval Process for New CTSA Program UM1 and K12 Projects that Involve Human Subjects Research
Process Overview
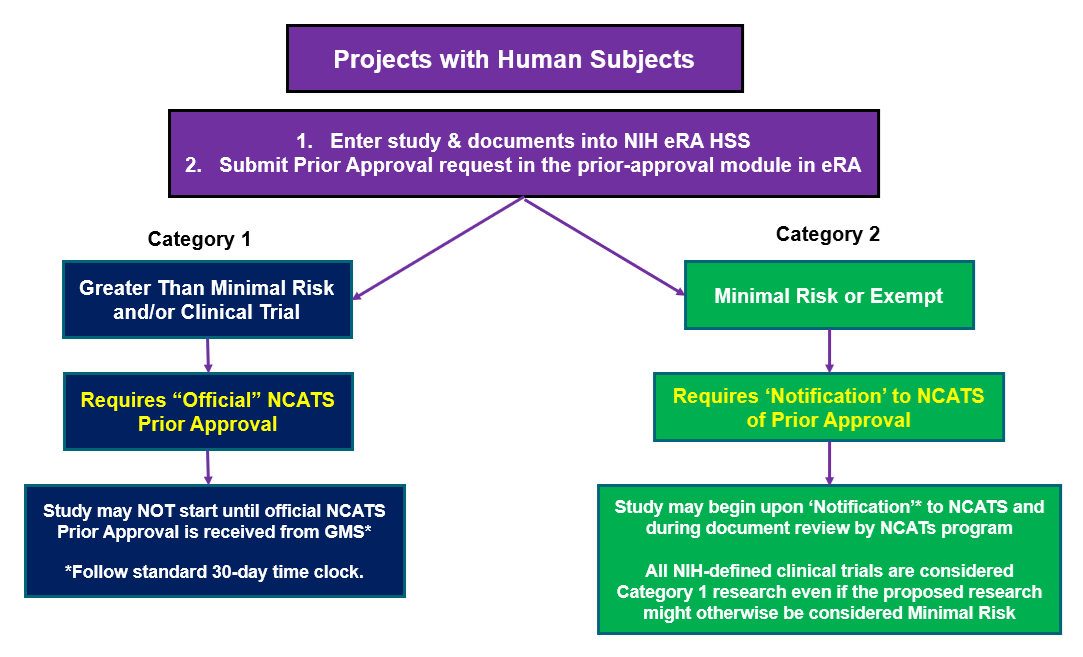
Category 1 Studies
Applies to: Projects involving human subjects deemed by the Institutional Review Board (IRB) to be Greater Than Minimal Risk and/or meet the criteria for an NIH-defined Clinical Trial.
Requires: Entry of the study into the eRA Human Subjects System (HSS) and a formal prior-approval request to the NIH prior-approval module.
An official notification from NCATS of NCATS’ Prior Approval before the project can begin is required.
Category 2 Studies
Applies to: Projects involving human subjects that are deemed by the Institutional Review Board (IRB) to be non-NIH-defined Clinical Trial minimal risk or exempt (45 CFR 46) studies.
Requires: Entries of these studies in the HSS along with NCATS-specified documentation and a and a formal prior-approval request to the NIH prior-approval module are required before the project can begin.
Note: NCATS does not require CTSA institutions to wait for NCATS approval to begin the study.
Instructions
A sample template for “NCATS NEW PROJECTS WITH HUMAN SUBJECTS RESEARCH” version 5 (Word - 198KB)
Submission of Prior Approval Requests
Please refer to CTSA Prior Approval FAQs for additional information.
Comments and questions about this process? Email us.
Requirement Reminders
- Documents must be uploaded to the NIH eRA HSS.
- Documents must be uploaded to the NIH prior-approval module.
- INFORMATION TO BE PROVIDED IN THE PRIOR APPROVAL MODULE UNDER “OTHER REQUEST”
- Description*: The HSS study title; HSS study PI; HSS#
- Effective Date*: The date the study is requested to start
- Justification*: Justification for delayed onset studies
- Budget: Leave empty; available in the HSS study record
- Other Supporting: Leave empty; available in the HSS study record


