CTSA Prior Approval Requests Frequently Asked Questions
The questions compiled here represent questions from the CTSA recipient institution community discussed in the CTSA Quality Assurance/Quality Control Group past meetings and questions posted in the QA/QC Group Discussion Forum.
Responses provided in this document are in accordance with NIH Grants Policy and do not supersede recipient institutional policies.
KL2/K12/TL1/T32 projects
Q: Should you include the KL2 scholar’s stipend in the NCATS prior approval request for a KL2 scholar Human Subjects System (HSS) or Vertebrate Animal Study (VAS) project?
A: The KL2/K12 scholar stipend or salary cannot be included with the KL2/K12 project budget. Please refer to section 2, question 2 of the Human Subjects Research Addendum.
Q: Should a KL2/K12 Scholar be appointed in eRA xTrain before completing the submission of a prior approval request?
A: Yes. Per the terms of award on all KL2/K12 grants, all statements of appointment must be submitted in xTrain before or at the start of each scholar appointment. If a HSS or VAS prior approval is submitted to NCATS before a statement of appointment is submitted in xTrain, NCATS approval will be delayed until the statement of appointment is submitted and accepted. No stipend or other allowance may be paid until the appointment form has been submitted and all grant provided research funds must go to grant supported scholars. It is also helpful if the Authorized Organization Representative (AOR)/Signing Official (SO) notifies the NCATS Grants Management Specialist (GMS) and Program Official (PO) once an appointment is submitted so they may begin the eligibility review.
Q: Can TL1 trainees be pilot project PIs?
A: The TL1 grant is a training mechanism for pre- and postdoctoral fellows preparing for a career in research and is not geared towards individuals engaging in independent research. However, in rare instances, TL1 trainees (particularly postdoctoral trainees) may conduct independent research supported through the UL1/UM1 Pilot Program Core. The CTSA Program hub must request prior approval for all research conducted by trainees through the Pilot Program that will be independent of the mentor’s research project and ensure proper oversight of the trainee-investigator.
Q: What is the (Inclusion Enrollment Report) IER requirement for KL2/K12 scholars who have their own Institutional Review Board (IRB) approval, but the research is also being supported by other grants/funds?
A: If the project is part of a larger NIH supported project that already has IER in place, there is no need to add IER for the KL2/K12 portion.
Q: How long does it take to review and approve xTRAIN appointments once submitted to NIH?
A: The review process should not take long, assuming there are no eligibility issues with the appointment (scholar and trainee eligibility requirements are included in the applicable Notice of Funding Opportunity [NOFO]). It is imperative that the recipient organization is aware of all awarded and pending grant applications for the trainee/scholar prior to appointment submission to ensure eligibility and avoid delay. Please note that the xTrain system does not notify the GMS or the grants management officer when the appointment is submitted in xTrain. Thus, it is recommended to notify GMS and program officer assigned to the grant when the appointments are submitted in xTrain. The notification will ensure there is NIH visibility on the pending appointment/termination and will speed up the review process.
Q: How are the IERs managed for a scholar that leaves the recipient institution? Is there a notation in the IERs indicating they are no longer going to be updated?
A: Recipients may put a comment in the IER comment area if they want to provide details on enrollment that they believe will be helpful (e.g., to indicate that enrollment stopped early because the scholar left the institution).
Q: What is the process for transferring projects from the KL2 to K12?
A: The KL2 termination notice and K12 statement of appointment must be submitted to NCATS in xTrain on the same day. This must be done before the project period end date of the KL2 award. The recipient institute AOR should then send an email to GMS and POs of the KL2 and K12 awards when the termination notice PHS 416-7 and statement of appointment PHS 2271 have been submitted with the following information:
- Names of the KL2 scholars that were appointed to the K12 in xTrain (VAS and HS).
- Titles of the scholar projects (VAS and HS).
- Associated study IDs (HS only).
- The official NCATS GMS prior approval for the KL2 scholars’ projects if transferring study is category 1 or VAS.
Please note that the NIH GMS does not receive notification when an appointment or termination is submitted, so email notification from the recipient institution is appreciated. This notification is not required to come from the AOR, however if there is additional information or documentation required to accept the appointment, AOR concurrence will be needed. Please refer questions to Patrick Brown.
Pilots
Q: If a study has several aims with different enrollment criteria per aim, should the PI provide several cumulative enrollment tables?
A: In general, for every planned enrollment report there should only be one cumulative enrollment table.
- Cumulative enrollment represents the cumulative record for all enrollment centers.
- The table should be updated with new records. In some cases, more than one IER for a study is acceptable: 1) when a study involves both an existing dataset/resource AND recruitment of new participants, or 2) when a study involves multiple, different existing datasets. Please also see “IV. Working with Existing Datasets and/or Resources.”
Q: A pilot recipient, whose study received NIH Prior Approval, is leaving the institution. The institution would like the study to continue, and they are working on identifying a PI to take over the study. How do we process this request through the HSS?
A: This should be discussed with the PO. The recipient institute would need to provide an updated IRB record with the new PI name, along with the HS training record for the PI. There is no need for a new prior approval if this is an ongoing pilot study, that has already received NCATS prior approval.
Note: If the PI of the pilot project is also a named key personnel on the parent UL1/UM1 Notice of Award (NoA), this would require a prior approval request on the parent UL1/UM1 grant submitted in accordance with NCATS’ Prior Approval guidance regarding a Change in Key Personnel. Generally, the named key personnel on the parent UL1 NoA are core directors and personnel that are considered essential to the execution of the award. The recipient should work with their AOR as they are well versed in this type of prior approval action.
Q: Is the HSS Section 3.1 an important part of the prior approval document and studies involving human subject research (HSR)?
A: It is an important part of the prior approval document. Please ensure that you follow the template provided in February 2022 QA/QC Meeting Presentation about HSS Section 3.1 and complete the section accordingly. This section should be completed to avoid delays in the prior approval process. Even if prior approval is not required, the recipient should ensure that HSS Section 3.1 is addressed, with all necessary sections explained in the February 2022 QA/QC Meeting. This document ensures that the study team is taking necessary steps to ensure participants’ safety and reduce risk.
Q: What is the reporting process for a pilot that needs to be extended to the next budget period?
A: All active HSS and VAS projects supported by NIH research funds and/or voluntary committed cost share must be reported in the Research Performance Progress Report (RPPR). Please reference the NCATS CTSA RPPR Supplemental Instructions for additional clarification. The pilot project activity may cross over budget periods. However, per NIH Grants Policy and the NoA, the institution cannot carry over funds from one budget period to another without NIH prior approval. Repeated prior approval requests to transfer funds from one budget period to another for the same/similar program costs will be denied. Please work with your Office of Sponsored Programs to make sure your pilot program follows NIH Grants Policy and avoids the need to set up carryover requests for pilot program funds.
Q: Why should recipients wait for two weeks after notification for category 2 pilots and K12/KL2 projects before starting the project?
A: NCATS POs will confirm that the HSS packet is complete, and the Category 2 designation is appropriate. A Category 2 pilot may start when all materials are submitted via HSS and the documents are being reviewed by NCATS staff, BUT if materials are not complete, are not clear, and/or the safety of the human subjects appears to be compromised in the study, the study may have to be paused while NCATS is acquiring additional materials. NCATS will communicate this to you through your AOR. Hence, NCATS recommends that the recipient waits two weeks before starting the study to allow NCATS to complete the internal review process.
Q: A recipient is waiting for prior approval. Is the recipient allowed to start working on the study now, using their own personal funds? If so, would the recipient be able to reimburse themselves with NCATS funding once prior approval is granted? The funding will be coming from direct grant dollars.
A: Under no circumstances may NIH-supported nonexempt HSR be initiated prior to obtaining IRB approval and providing the final IRB approval date to NIH. NIH will not allow recipients to use any funds if a certification and an IRB approval date have not been provided. NCATS discourages using mixed funds to perform studies in line with the following:
Recipient organizations are not permitted to mix and match sources of funding that have different prior approval requirements. For example, recipient organizations should not partially fund a pilot project with direct funding and voluntary, uncommitted cost sharing.
Source of Pilot Funds | Prior Approval Required |
Direct Funding | Yes |
Voluntary Committed Cost Sharing Note: UM1 and K12 projects can only be funded with NCATS direct funding. | Yes for UL1 and KL2 |
Voluntary Uncommitted Cost Sharing | No |
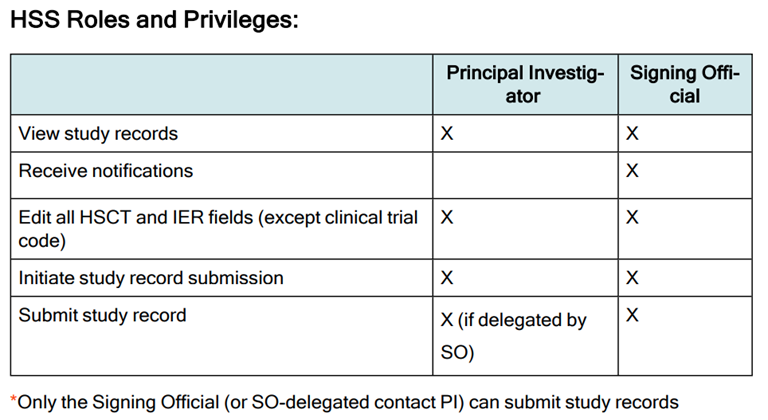
Screenshot of Roles and Privileges Section of the Human Subjects System (HSS) for Institution Staff User Guide
Q: For category 1 study (greater than minimal risk and/or clinical trial), if we don’t hear back from the NCATS prior approval within 30 days, should the recipient reach out to NCATS?
A: Yes. If a recipient does not hear back from the NCATS prior approval within 30 days, they are encouraged to reach out. The recipient also can reach out to the Program Officer with any questions related to the prior approval process.
Q: For category 2 studies, does the AOR need to submit both the submission in HSS and submit the notification/request?
A: All HSS submissions must have AOR concurrence. This can be accomplished in two ways: Concurrence is indicated via the AOR submitting the study request in HSS or if the AOR delegates HSS submission roles within the HSS system the AOR must submit the notification/request clarifying the AOR role to the NCATS Prior Approval Mailbox copying the assigned Program Officer and GMS.
Clinical Trials
Q: A recipient has submitted a clinical trial prior approval request. Is it allowable to begin recruitment without completing study procedures until NIH approval?
A: If the recipient did not receive a formal approval of the prior approval submission, no interaction with study participants is allowed. Only administrative activities are allowed. The recipient should reach out to the PO to discuss. In general, it should not take more than 30 days for a prior approval to be officially approved by NCATS.
Q: What are the Reporting Requirements to clinical trials.gov for clinical trials for applicable clinical trial (ACT)* and NIH-Funded Clinical Trials**?
A: Please see NIH’s guidance on Clinical Trial Requirements for Grants and Contracts.
HSS Study Close Out
Q: How can I inform my PO that my study is completed?
A: There is no NIH-wide “close-out” process for study records. However, be aware of any NOFO-specific requirements. NIH expects recipients to:
- Clearly describe study information on PHS human subjects and Clinical Trials Information Form.
- Keep study status and key study dates up-to-date and accurate.
- Describe study status in progress report, as appropriate.
- It is required to upload clinical trial result information to CT.GOV in compliance with NIH Grants Policy Statement, Section 4.1.3.1. within 1 year of completing clinical trial enrollment.
For more guidance, please refer to the October 2022 QA/QC Meeting on “Documenting Study Activities in the Human Subjects System,” presented by Dawn Corbett, M.P.H., NIH inclusion Policy Officer from the Division of Human Subjects Research, Office of Extramural Research. View the presentation slides and video.
Q: What are the reporting requirements for pilots that received NCATS funding and continue the study beyond the NCATS funded period, with nonfederal funding?
A:Once pilots complete their NCATS funded period, no reporting is required in the RPPR or HSS system. Once a KL2/K12 scholar terminates their appointment, no further reporting is needed in the RPPR or HSS.
All clinical trials must ensure they are compliant with NIH clinical trial reporting requirements regardless of the duration of the award.
QA/QC Administration
Q: How can a QA/QC personnel get access to the discussion forum and all QA/QC resources?
A: To access the discussion forum and all QA/QC resources:
- Visit the QA/QC Group Discussion Forum.
- View the Instructions for Participating in the QA/QC Discussion Forum (PDF - 637KB)
If you have questions or require assistance, contact Lenore Roca.
All past QA/QC resources (meetings, FAQs, etc.) can be found on the QA/QC Group Shared Drive (login required). If you have difficulty accessing the shared drive, please request help from email support.


